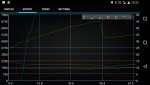
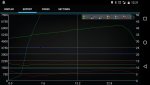
Humidity plays a huge role in my IAT. Also the more meth vs water = less cooling
TL;DR
Not completely true. In the combustion chamber, yes, water cools more. In the air stream? Depends. Humidity temperature delta and time precict the winner. For most applications, higher percent of alcohol or pure alcohol wins.
TL;DR#2
Use as much alcohol as you deem safe. The safest is 49/51. Best benefit vs safety is probably 70/30. 100% alcohol is for sailors and racers or people who aspire to be both.
.......
Water has a better latent heat of vaporization, but it is harder and slower to evaporate. Pure alcohol evaporates very fast even in humid conditions. Evaporation is where the cooling magic happens and there is only so much time to do it.
Temperature difference, time and humidity.
We have an intercooler. Even maxed out on hot day with a tuned car, it's still doing some decent work. At red line with a tune you might be pushing something near 400f degree air out of your turbo. That gets knocked down to 140f by the intercooler.
Now we're working with the difference between 140 and ambient.
If ambient is 80, the difference = 60 degrees. With a delta of only 60 degrees and humidity, water can only evaporate so much and in so much time. Most of the water will pass right through and only evaporate in the combustion chamber.
The water cooled your combustion process. But at the cost of taking up space with non combustible material and your intake didn't see the cooling.
If we ran no intercooler and the water was fighting 400 degree air, it would evaporate more before the combustion chamber and contribute more cooling effect in the air stream. A 320 degree delta offers more evaporation evaporation driving force than 60.
With the small temp delta we're working with + humidity, alcohol evaporates lightning quick, gives you all its latent heat of vaporization in the air stream, and ads octane + combustion power to the process.